RICHLAND, Miss.—At 17, Christopher Pena has spent his life battling Barth syndrome, a rare genetic disorder that weakens the body. The Rankin County, Mississippi, teen recently learned the drug he credits with giving him strength to jump, carry heavy items and keep up in school won accelerated approval from the Food and Drug Administration.
The FDA granted clearance for elamipretide, which will be sold under the brand name Forzinity, as the first treatment for Barth syndrome in patients over 30 kilograms—about 66 pounds.
The decision came during uncertainty surrounding continued access to the drug. In May, the agency denied the drug’s application from Boston-based Stealth BioTherapeutics, which is developing therapies for Barth syndrome and other mitochondrial diseases. Since then, Christopher and his family had hoped the FDA would reconsider, especially after his year on the medication showed noticeable improvements.
‘I Realized I Could Jump’
Before starting Forzinity last fall, Christopher said his days were shaped by exhaustion and the constant threat of heart failure, seizures and strokes—hallmarks of Barth syndrome.
“It was a dream to have a medicine that would help me,” he said.
Lindsay Marjoram, director of research for the Barth Syndrome Foundation, said the disorder attacks the body’s energy supply, damaging the heart, skeletal muscles and immune system.
“The heart’s bigger than it should be, but it’s also weak,” she said. “All the other muscles in your body that you use to walk and run—they also don’t make enough energy—and so there’s kind of an exercise intolerance, and what that ultimately does is it makes it really hard for somebody who has Barth syndrome to participate in life.”
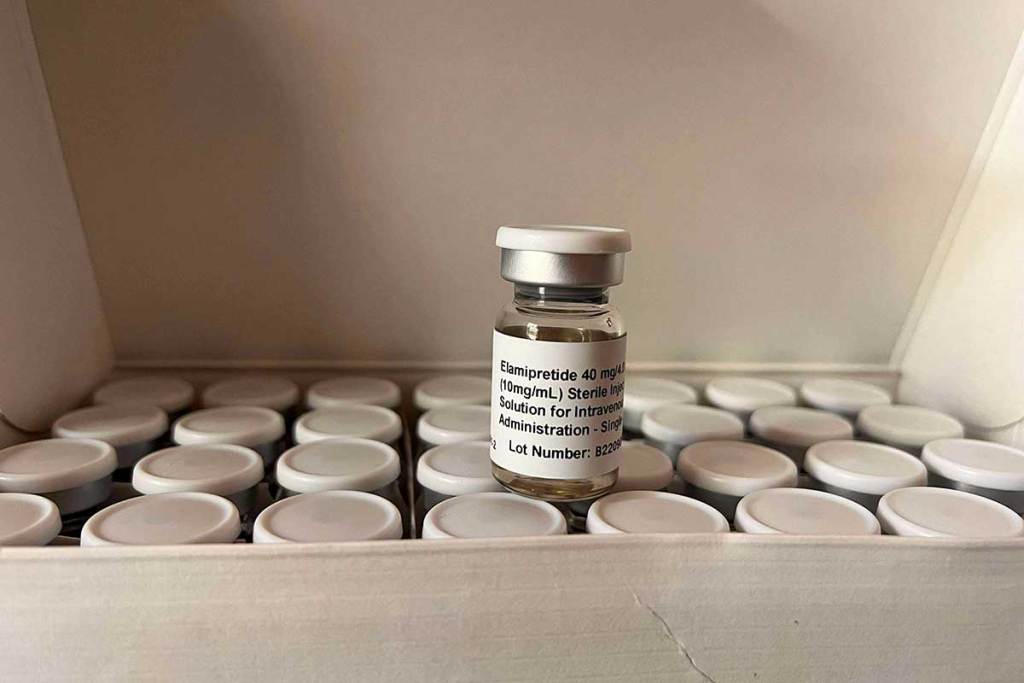
Forzinity, which was in development for more than eight years, is designed to target the part of cells that produce energy, boosting stamina for people with Barth syndrome.
Within months of starting treatment, Christopher said he could do simple things he had always struggled with.
“Honestly, I started getting more energy,” he said. “But when I really noticed it, I was out in my backyard playing with my dog and started jumping—and realized I could jump—which was something I could never do before.”
In the wake of the decision, Christopher hopes for a future not dominated by his health.
“I’ll be able to go out, have friends, have a social life, go places with friends, go places with family,” he said. “Be able to go to school, work, where before I wouldn’t … have the energy or the strength to do that stuff.”
Next Gaps to Close
Christopher’s mother, Kristi Pena, said the months before the FDA’s decision were dominated by uncertainty. She traveled to Washington, D.C., in late August to press officials for action.
“As a parent who’s had a child born into a world that (was) told, ‘There’s nothing we can do to help them,’—that’s a hard, bitter pill to swallow,” she said. “Then we learned of this drug, and that’s something we can do, and we fought for it, and now it’s here.”

Her fight coincided with a shift inside the agency. On Sept. 3, the FDA rolled out new guidelines aimed at speeding reviews of drugs for ultra-rare conditions such as Barth syndrome. The Rare Disease Evidence Principles allow more flexible trial designs and encourage earlier communication between regulators and drugmakers. Forzinity’s accelerated approval follows that framework, resting on a functional strength measure and a confirmatory trial soon to begin.
Kristi said the approval turned fear into logistics: transitioning from expanded access to a prescription, navigating insurance and ensuring uninterrupted supply as specialty pharmacies begin dispensing Forzinity.
“That’s at the forefront of our mind, and that’s definitely an obstacle we’re going to have to overcome,” she said. “But I believe that we have the right people advocating for us on both sides … and I believe with all of them combined with parents who are like us, who just don’t take no for an answer, that will have a positive outcome.”
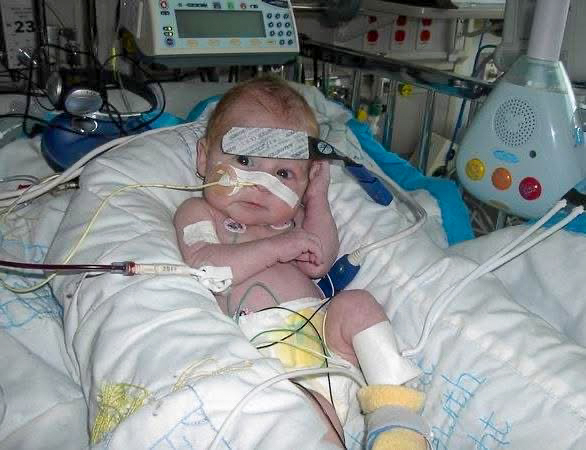
Beyond insurance concerns, Lindsay Marjoram said another priority is closing the gap for children under 66 pounds, given that about 40 percent of children with Barth syndrome die by age 5. The drugmaker is working with the FDA to produce data that would expand access.
“For so many years, it’s been tenuous whether this drug was ever going to get approved, and people have seen others improve when they’re on this drug, and now there’s real hope,” she said. “It’s tangible, and I do hope that it opens the door to future drugs being developed so that the entire community can benefit and no longer have their lives run by Barth syndrome.”
For Christopher, that hope translates into new goals—some that once seemed out of reach.
“We’ve been fighting for this medicine since I was 6, 7 years old, so it’s a big deal to get it now,” he said. “I’ve always wanted to go to college, and that’s never really, truly been an option … but now that’s something I’ll be able to do.”
This article first appeared on RHCJC and is republished here under a Creative Commons Attribution 4.0 International License.






